APEX® Technology
Protection against Corrosion
Acid-Resistant Heat Exchangers Constructed from Polymer Composite
Polymer Composite PPS-Gr
For constant operation below the Acid Dew Point (ADP), APEX Group developed the heat exchanger constructed of polymer composite, resistant to highly concentrated acidic condensate.
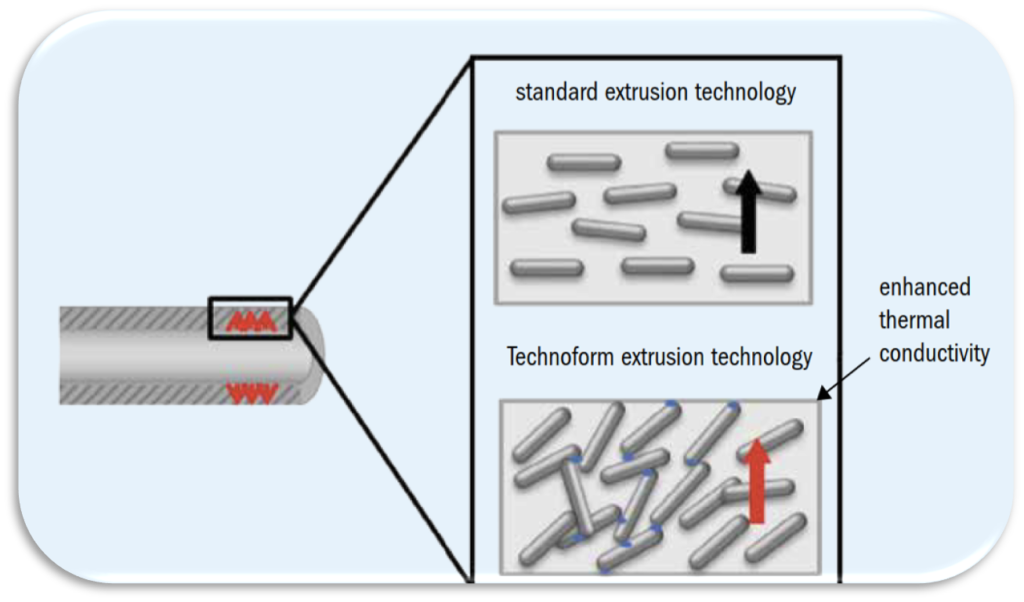
Polymers offer excellent resistance to chemical attacks. However, the drawback of most polymers is their low thermal conductivity, and their consequent thin walls required for an efficient heat transfer. This drawback results in a lower mechanical strength of the heat transfer elements.
A polymer composite overcoming this drawback is PPS Gr. The PPS Gr composite consists of polyphenylene sulphide and graphite (a heat-conductive filler), and thus features resistance to chemical attacks and additionally high thermal conductivity. The heat transfer walls can be designed thicker in order to improve mechanical strength, overcoming the thermal resistance of the polymer.
Background: Formation of H2SO4 (gaseous or liquid fuel firing) and the Working principle

The conversion of SO2 to SO3 is a complex process depending on many variables, e.g.:
- Fuel Sulphur content and fuel composition;
- Excess air;
- Combustion and mixing process (gas kinetics);
- Temperature and pressure pattern in the system;
- Flue gas residence time in the system;
- Presence of catalysts (solid surfaces or flue gas components);
- Soot and other solid particles in the system
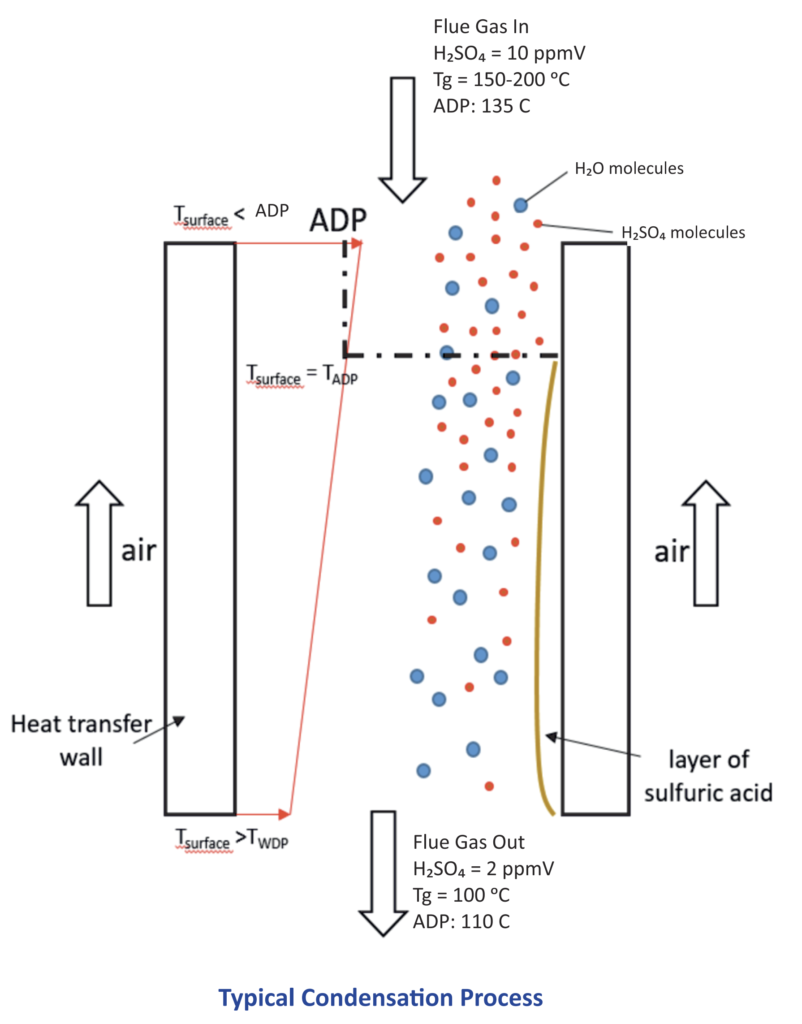 The flue gas temperature at the channel inlet is usually above the ADP and all its components form a homogeneous gaseous phase. Immediately after the surface temperature on the flue gas side drops below the ADP, H2SO4 (g) starts condensing on the surface and building a highly corrosive liquid acidic film on the surface [H2SO4 (l)] leading to a reduction of H2SO4 concentration of the flue gas.
The flue gas temperature at the channel inlet is usually above the ADP and all its components form a homogeneous gaseous phase. Immediately after the surface temperature on the flue gas side drops below the ADP, H2SO4 (g) starts condensing on the surface and building a highly corrosive liquid acidic film on the surface [H2SO4 (l)] leading to a reduction of H2SO4 concentration of the flue gas.
The liquid film will grow in time (growth rate depending on the concentration of H2SO4 (g) in the flue gas).
At flue gas bulk temperatures below the ADP, H2SO4 may condense on particles (nuclei) existing in the flue gas (heterogeneous condensation) and /or spontaneously as homogeneous condensation, in which case the condensation rate on the surface is diminished.
At the outlet of the heat exchanger, a corresponding portion of H2SO4 remains in its gaseous state and is released as H2SO4 (g) out of the heat exchanger.
The amount of H2SO4 entering as a homogeneous phase is split inside the channel into 3 distinct portions which, at steady state, leave the channel as follows:
Portion 1: H2SO4 (g) in the flue gas bulk stream; this is the portion which determines the ADP at the channel exit;
Portion 2: H2SO4 (l) formed as film on the surface;
Portion 3: H2SO4 (l) as acid mist in the bulk stream.
The distribution between these three portions depends on the operating conditions.
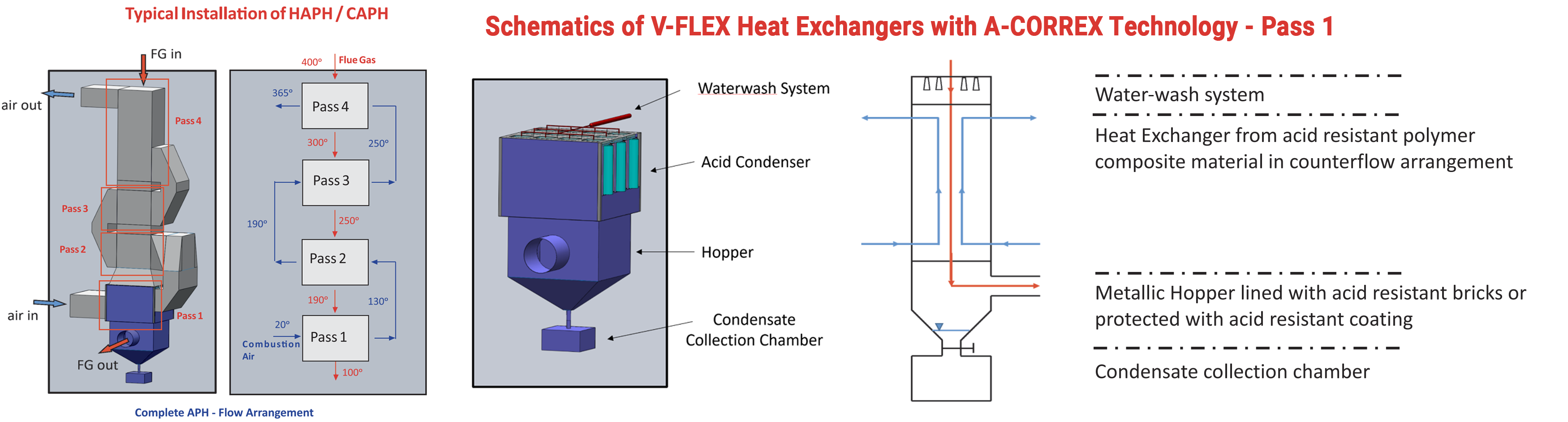
Key Benefits
- Resistant to virtually any chemical attack below ADP and WDP
- Relatively high thermal conductivity comparing to pure polymers (> 3 W / m-1 K-1)
- Operating temperature up to 200 ° C (short term peaks 220 °C)
- Good resistance to heat cycles and thermal shocks
- Modular to easily adapt to different operating conditions
Typical Applications:
- DeNOx or DeSOx Gas/Gas Heater
- “cold-end” Air Preheater
- CCS Flue Gas Cooler
A-CORREX Technology for both Plate and Tubular Acid-Resistant Heat Exchangers
1. PPS Gr Flat Plate Heat Exchangers: Compact and Energy-Efficient
Characteristics:
- Compact and integrated with the hot metallic passes;
- Light weight.

2. PPS Gr Tubular Hear Exchangers: Cost and Time Efficient
Characteristics:
- Allowing off-line replacement of individual modules;
- Cost and time efficient.

